Batch report — full panel
RET-20-V-2604-001
Retatrutide vial line (10mg and 40mg formats) — the only batch in this catalog with the full sterility-grade panel: 99.741% HPLC, 20.92 mg assay, heavy metals not detected, TFA not detected, bacterial endotoxin 1.449 EU/Vial. Each report has its own Janoshik verification key.
For in-vitro laboratory research only. Not for human or veterinary use.
Batch facts
- Products
- Retatrutide 10mg Vial · Retatrutide 40mg Vial
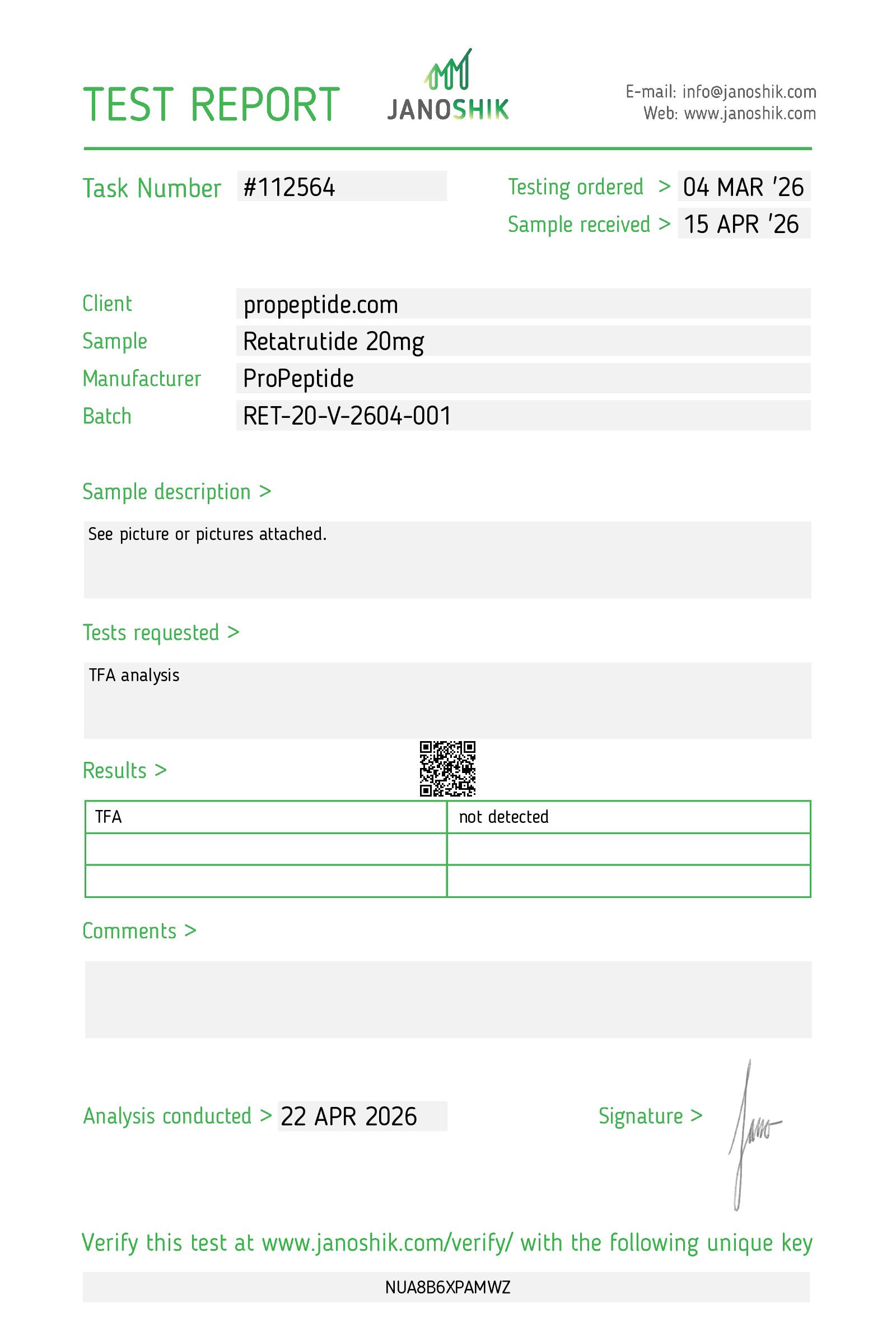
- Batch number
RET-20-V-2604-001- Molecule
- Retatrutide (LY-3437943) — CAS 2381089-83-2
- Purity (HPLC)
- 99.741%
- Assay
- 20.92 mg retatrutide
- Heavy metals
- As, Cd, Pb, Hg — not detected
- TFA
- Not detected
- Endotoxin (LAL)
- 1.449 EU/Vial
- Laboratory
- Janoshik Analytical
- Analysis dates
- 21–24 April 2026
- Verification keys
- Purity
YCYA75M373NA· Heavy metalsGHHWTCPUSEZ7· TFANUA8B6XPAMWZ· EndotoxinE8ZFE17MI47D
Heavy metals report
ICP-MS panel for arsenic, cadmium, lead and mercury. All four below detection limits.

TFA report
Trifluoroacetic acid is a residual solvent from peptide synthesis. HPLC purity does not see it. This batch tests separately and returns "not detected."
Common questions
What is batch RET-20-V-2604-001?
Retatrutide vial line covering both 10mg and 40mg formats. Janoshik ran a full panel 21–24 April 2026: 99.741% HPLC, 20.92 mg retatrutide assay, heavy metals (As, Cd, Pb, Hg) not detected, TFA not detected, bacterial endotoxin 1.449 EU per vial.
What does 1.449 EU per vial mean?
EU is endotoxin units, measured via the LAL (Limulus Amebocyte Lysate) assay. 1.449 EU per vial is the bacterial endotoxin load found in the vial. Endotoxin is the sterility benchmark pharma uses for release testing. Most research peptide sellers skip it; this batch publishes the number.
Why test for TFA separately?
TFA (trifluoroacetic acid) is a common residual solvent from peptide synthesis. HPLC purity does not see it. Testing separately confirms it is not present. For this batch, TFA was not detected.
How do I verify each Janoshik report?
Go to janoshik.com and paste the relevant verification key. Purity:
YCYA75M373NA. Heavy metals: GHHWTCPUSEZ7. TFA: NUA8B6XPAMWZ. Endotoxin: E8ZFE17MI47D. Each returns the original report off Janoshik's server.Source these vials from Remy Peptides
Both the 10mg and 40mg vial SKUs ship under batch RET-20-V-2604-001 while it remains the current vial batch.

